Effect of non-steroidal antiandrogens on sexual interest and function: Exclusive Research!
Hello dear readers! We conducted an exclusive research on sexual health and want to share this information with you. Only with us! Exclusive article.
Effect of non-steroidal antiandrogens on sexual interest and function Nilutamide Published clinical experience with nilutamide monotherapy is limited to a single open uncontrolled study in which 26 patients with metastatic prostate cancer were treated with nilutamide 100 mg three times daily and studied for 10 months. Sexual potency in this
study was assessed by patient interview only. 
Fifteen of the men claimed to be potent before treatment, of whom seven (47%) maintained libido and potency on nilutamide; libido and potency were completely abolished in five patients (33%) and attenuated in the remaining three (20%). Of the seven men in whom potency was maintained on nilutamide, all four who were subsequently switched to a luteinizing hormone-releasing hormone (LH-RH) agonist experienced a loss of libido and erectile function.
Flutamide
Flutamide has been used more widely as a monotherapy for advanced prostate cancer than has nilutamide. The majority of published data are from relatively short, small, open studies, involving patients with metastatic disease. Only one small (n=104) study of flutamide monotherapy versus castration has been published33, in which median time to
progression and overall survival at 69 months were similar for the two treatments. There are no comparative data on the effects of treatment on sexual parameters, as the majority of patients were impotent before study entry. Three small pilot studies have also investigated the combination of the 5α-reductase inhibitor finasteride with flutamide.
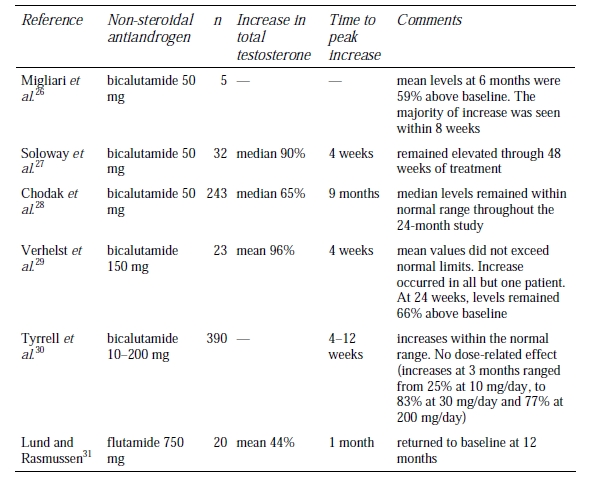
This combination resulted in a mean reduction in serum prostate-specific antigen (PSA) levels of > 90%. Details of changes in sexual interest and/or function in these studies and in trials of flutamide mono-therapy are presented in Table 1 data were obtained by patient self-reporting or direct patient questioning with the exception of the study by Brufsky’s group, which used a patient-completed questionnaire. In several of these studies, around two-thirds of previously sexually potent men retained potency during treatment with flutamide; a small proportion of previously impotent men regained sexual function on flutamide.
There are fewer data on sexual interest, but it appears to be preserved in a similar proportion of patients. The results of the large randomized study of Schröder and colleagues (European Organization for Research and Treatment of Cancer (EORTC) protocol 308929) are a notable exception to the other results presented in Table 1. This study compared flutamide and the steroidal antiandrogen cyproterone acetate (CPA) in M1 prostate cancer patients with good prognostic factors.
The authors found that the decreases in sexual function and activity with flutamide were surprisingly large (Table 1), and although less pronounced, the decreases were not statistically different from those observed with CPA. Contributing to the apparent discrepancy could be the long follow-up in the EORTC study (in excess of 2 years).
Furthermore, the patients receiving flutamide were significantly older with a median age of 73 years versus 69 in the CPA group. Still, these results are unexpected given that CPA exhibits pronounced progestogenic effects, thereby reducing gonadotropin secretion and testosterone synthesis. Consequently, CPA has previously been reported to be
associated with a severe suppression of libido and loss of erectile potency, the incidence of which is comparable to that seen following castration.
Flutamide has also been investigated in benign prostatic hyperplasia (BPH). Results on changes in sexual function in these studies confirm the findings in advanced prostate cancer patients. In a double-blind, placebo-controlled, dose-response study involving 372 men aged 42–88 years (mean 66 years) with BPH62, sexual dysfunction was reported by a small proportion (3–8%) of each treatment group after 24 weeks, with no significant difference between the groups. In a smaller (n=87), open, placebo-controlled study, none of the BPH patients (mean age 65 years) reported impotence after treatment with flutamide 750 mg/day for periods of up to 6 months.
Table 1 Summary of data on sexual interest and function in published studies of flutamide monotherapy for advanced prostate cancer.
Studies using bicalutamide 50 mg/day

In the initial studies of bicalutamide monotherapy in metastatic prostate cancer patients, a 50-mg daily dose (the dose otherwise used as a component of CAB) was compared in an open manner with castration (surgical or medical). Since these open, randomized, multicenter trials had a similar design, pooling of the data was possible. An overview analysis of these studies involving a total of more than 1000 patients with metastatic prostate cancer revealed that there was a median survival difference between the treatments of approximately 3 months, favoring castration14. Therefore, in subsequent studies in prostate cancer patients, bicalutamide monotherapy was evaluated at 150
mg/day, rather than at 50 mg/day.
In all three studies in which the bicalutamide 50-mg dosage was used, quality of life was assessed using a self-administered questionnaire. When the data were pooled, the scores for sexual interest and function were superior in the bicalutamide group. In the largest of the studies involving 486 men, sexual interest and function were maintained with bicalutamide for up to 3 months, with a slight decline thereafter. In contrast, in the castration group there was a decline in both parameters within 1 month and there was a significant difference between the treatments, favoring bicalutamide 50 mg/day, at 1, 3 and 6 months for sexual interest and at 1 and 3 months for sexual function.
Sexual function has also been assessed objectively in patients receiving bicalutamide 50-mg/day monotherapy. Using continuous monitoring of penile tumescence and rigidity on multiple nights in five men (mean age 61 years) with advanced prostate cancer, Migliari and associates26 found that 6 months’ therapy with bicalutamide 50 mg/day had no effect on the number of tumescence episodes, tumescence duration, maximal change in penile circumference or duration of rigidity. Only one of the patients (20%) reported a decrease in sexual desire and libido. Bicalutamide 50-mg/day monotherapy has also been evaluated in three double-blind, placebo-controlled studies in a total of 126 BPH patients over 24 weeks64,65 (data on file).
Although these studies used different questionnaires to assess sexual parameters, findings were consistent across the studies, with all three concluding that retention of both sexual interest and function was comparable in the bicalutamide and placebo groups; these data are in agreement with the findings on flutamide in BPH. A further analysis of data from the largest of these studies in patients with high sexual interest and high sexual function at baseline showed that the impact of bicalutamide on both parameters was similar to that of placebo (data on file). It is interesting to note that even with placebo there was a decline in sexual interest and some loss of sexual function in these patients over time.
Studies of bicalutamide 150 mg/day
Subsequent bicalutamide monotherapy studies were conducted using 150 mg/day. Two large phase III studies comparing bicalutamide 150-mg/day monotherapy with castration (orchidectomy or the LH-RH agonist goserelin acetate) in 1453 patients with locally advanced (M0) or metastatic (M1) prostate cancer had an identical design, to permit a combined analysis. In the pooled analysis of 480 M0 patients after a median follow-up of 6.3 years with 56% deaths, there was no overall difference in survival or time to progression with bicalutamide 150 mg/day compared with castration.
Two smaller open European studies have compared bicalutamide 150 mg/day with CAB in patients with advanced disease. The study population (n=220) of Boccardo’s group48 comprised approximately equal proportions of locally advanced and metastatic patients, whereas the 235 patients enrolled by Chatelain and colleagues66 mainly had metastatic disease at entry. Survival data in these trials are not currently mature, but indicate that CAB is not more effective than bicalutamide 150-mg/day monotherapy.
Quality of life, including sexual interest and function, was an important outcome measure in all four of these phase III trials, and these studies attempted to overcome some of the difficulties in assessing sexual function described above. Several analyses focused specifically on patients with sexual interest and/or function at study entry. In addition, although there is no universally accepted health-related quality of life instrument for prostate cancer patients, all four phase III studies used the same validated questionnaire, covering ten domains including sexual interest and function9, which was completed in private by the patient, although Boccardo’s group48 also used direct questioning.
As described above, the two largest monotherapy studies of bicalutamide 150 mg/day had an identical design to permit a pooled analysis. Separate analyses of quality of life data were performed at 12 months for locally advanced and metastatic patients. Of the 480 patients with locally advanced disease at entry, 337 responded to questions
on changes in sexual interest during the 12-month period. The combined analysis at 12 months showed that bicalutamide was associated with a significant advantage for sexual interest compared with castration (p=0.029), although a decrease was recorded in both groups11 (Figure 1). The reduction in the mean score for the sexual interest dimension was 23% for bicalutamide 150 mg/day and 47% for castration.
Figure 1 Percentage reduction from baseline in sexual interest after 12 months’ treatment: bicalutamide (Casodex®) 150 mg daily versus castration in patients with locally advanced M0 prostate cancer.
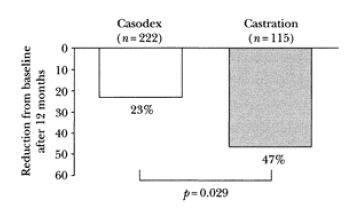
After the first month of treatment, the decrease in sexual interest was slightly greater in patients receiving castration. After that, patients treated with castration experienced a further decline, unlike patients receiving bicalutamide. This is consistent with the fact that following medical castration (which was used in 86% of the castration group) testosterone concentrations, after an initial increase (flare), decrease to within the castrate range within 21 days. A dramatic and sustained fall in sexual interest follows the reduction in testosterone concentrations. In contrast, the reduction in sexual interest with bicalutamide 150 mg/day occurs after the diagnosis has been established and changes little over time. Thus, the benefits of bicalutamide 150 mg/day over castration are evident within a month and are maintained for at least 12 months.
Approximately 50% of M0 patients in both treatment groups had no sexual interest at study entry. Further analyses were therefore performed on data (last value carried forward (LVCF) analysis) provided by only those patients with some degree of interest at baseline (i.e. those who had scope for deterioration). This allowed assessment and
comparison of changes in sexual interest of the two treatments.
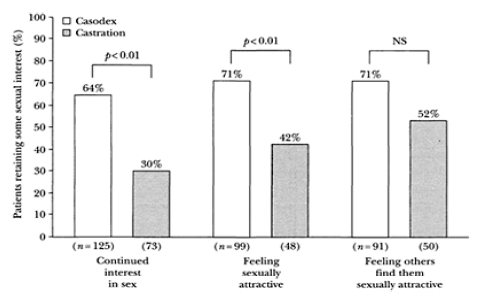
Since it takes some time for castrated patients to lose their interest in sex, using this type of analysis was not expected to produce any bias against castration. In this exploratory analysis, the benefit for bicalutamide 150 mg/day was even more pronounced than in the overall study population. Of those patients with sexual interest at study entry who responded to the specific question on sexual interest (n=198), 64% of the bicalutamide group still had some interest in sex during the 12 months compared with only 30% of the castration group (p<0.01). Similarly, 71% and 42% of the bicalutamide and castration groups, respectively, still felt they had some sexual attractiveness (p<0.01; n=147). Patients receiving bicalutamide 150 mg/day as monotherapy were also more likely still to feel that
others found them to have some sexual attractiveness (71% vs. 52%; n=141), although the difference did not reach statistical significance (Figure 2).
Figure 2 Sexual interest of patients with locally advanced M0 prostate cancer after 12 months’ treatment: bicalutamide 150 mg versus castration. NS, not significant
Similar results were obtained when patients with metastatic disease at study entry were analyzed, although the differences between the bicalutamide 150-mg/day and castration treatment groups were not as great as for the patients with less advanced disease10 (data on file). At 12 months, data from the 210 men answering questions on the sexual interest dimension showed that sexual interest was maintained to a significantly greater extent with bicalutamide 150 mg/day than with castration (p=0.041), with a difference between the groups apparent at 1 month.
As with the locally advanced disease group, a significant proportion of patients (56%) had no sexual interest at study entry. In exploratory analyses, excluding these patients (data on file), there was a clear difference (p<0.01) between treatment groups with respect to the proportion of patients retaining some sexual interest (67% vs. 40%; LVCF analysis). Differences were also apparent for feeling sexually attractive and feeling that others found them sexually attractive, although these were not significant.
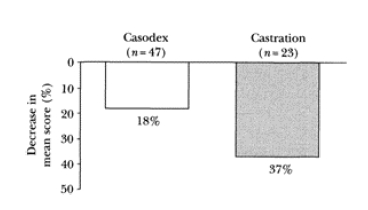
A smaller number of patients in these two studies answered questions on sexual function at 12 months, and statistical analyses could not be undertaken. In the locally advanced patients (n=70), sexual function was reduced by 18% from baseline in the bicalutamide 150-mg/day group and by 37% in the castration group68 (Figure 3). The
reduction in sexual function in the castration group was lower than expected, and this may have been because patients who lost sexual function were less likely to answer the relevant question (which was asked after the question on sexual interest) than the minority of patients who had some sexual function remaining.
The results of the two phase III studies of bicalutamide 150-mg/day monotherapy versus CAB support the above findings. In the study of Boccardo’s group, of those responding to direct physician enquiry who had normal libido and erections at entry, significantly more patients treated with bicalutamide 150 mg/day retained sex ual interest during follow-up (median 38 months) than in the CAB group (40% vs. 15%; p=0.01).
Figure 3 Percentage reduction from baseline in sexual function after 12 months’ treatment: bicalutamide 150 mg versus castration in patients with locally advanced M0 prostate cancer.
Similarly, the proportion of patients who maintained their erectile capability was greater in the bicalutamide 150-mg/day group than in the CAB group (31% vs. 7%; p=0.002). Responses to the quality of life questionnaire, which was completed by 130 men, showed significant (p≤0.05) differences for sexual interest, favoring bicalutamide, at 2, 6 and 9 months. In the other study66, there was also a non-significant trend in favor of bicalutamide 150 mg/day with respect to sexual interest over the first 24 months of treatment.
As mentioned above, the interpretation of data on sexual interest and function from open studies in which castration is a comparator can be difficult. However, data on bicalutamide have also been obtained from two blinded studies (data on file), the results of which are consistent with those described above. In the first of these, the impact of bicalutamide on sexual interest and function was evaluated using the EORTC quality of life questionnaire in a randomized, double-blind phase IIb study versus chlormadinone acetate 100-mg/day monotherapy in 109 Japanese patients with locally advanced (36%) or metastatic (64%) prostate cancer (data on file);
Chlormadinone is a steroidal progestogen with antiandrogenic properties, widely used for the treatment of prostate cancer in Japan. In this study, the daily dose of bicalutamide was 80 mg, which, based on weight-adjusted plasma bicalutamide concentrations, is a dose pharmaco logically equivalent to 150 mg in Western patients (data on file). Of the bicalutamide-treated patients with some sexual interest at study entry (n=28), 64% retained sexual interest and 77% maintained sexual function after 3 months’ treatment.
Bicalutamide 150 mg/day was also compared with flutamide 750 mg/day in a randomized single-blind study involving 148 patients with locally advanced disease (data on file). Quality of life was assessed using the Rotterdam symptom questionnaire, which includes a question on sexual interest. At 12 weeks, 72% of bicalutamide patients and 68% of flutamide patients showed no worsening in their responses concerning sexual interest, compared with baseline. This result for flutamide is consistent with previously published data (Table 1).
The Scandinavian Prostatic Cancer Group study 6 (SPCG-6) is part of the large bicalutamide Early Prostate Cancer Program, and included a total of 1218 men with early non-metastatic prostate cancer, of whom slightly more than 80% had not undergone therapy of primary curative intent (i.e. were otherwise candidates for watchful waiting). Patients were randomized beween bicalutamide monotherapy 150 mg daily and placebo. A first preliminary analysis of time to progression is just about to be published.
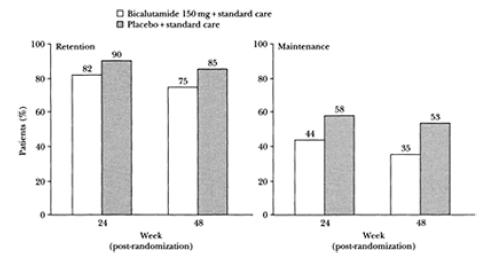
Sexual function was assessed using a shortened version of the Golombok and Rust Inventory of Sexual Satisfaction (GRISS) questionnaire, which was completed by the patient at baseline and at 12, 24, 36 and 48 weeks after randomization (Table 2). This questionnaire comprised six questions, two addressing the frequency of sexual activity and four concerning erectile function. The questions were answered in a semiquantitative manner (never, rarely, some times, etc.), allowing a numerical score to be assigned for the purposes of analysis. The proportion of men retaining some sexual activity and function (which could be less than at baseline) and the proportion maintaining sexual activity and function (the same or higher than at baseline) were calculated.
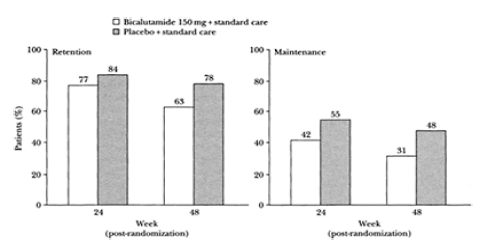
The results of the GRISS questionnaire obtained at 24 and 48 weeks after randomization are presented in Figures 4 and 5. At 48 weeks, 78% of patients in the placebo group retained some sexual activity as opposed to 63% in the bicalutamide group. Corresponding figures for maintenance of sexual activity were 48% and 31%. With respect to sexual function, 85% of placebo patients retained some sexual function at 48 weeks compared with 75% of patients in the bicalutamide arm, while 53% and 35%, respectively, maintained their pretreatment level of sexual function. Thus, while a higher proportion of patients retain and maintain sexual activity and function after randomization to placebo than to bicalutamide, the differences are relatively small, with the majority of patients retaining some sexual activity and function after treatment with bicalutamide.
Table 2 The GRISS (Golombok-Rust Inventory of Sexual Satisfaction) shortened version questionnaire used in the Scandinavian Prostatic Cancer Group study 6 (SPCG-6)70 to assess sexual activity (questions 1 and 2) and sexual function (questions 3–6). Possible answers are: never, hardly ever, occasionally, usually, always.

Conclusions
Quality of life issues are increasingly influencing treatment choices in prostate cancer patients, and sexuality is an important component of quality of life for many middle-aged and older men. Sexual interest is a prerequisite for sexual activity and cannot be effectively treated, whereas in patients with sexual interest, erectile dysfunction can be managed in a variety of ways. Lack of sexual interest, therefore, should be regarded as a greater problem than erectile dysfunction.
Figure 4 Sexual activity as assessed by the GRISS (Golombok-Rust Inventory of Sexual Satisfaction) questionnaire in men treated with bicalutamide 150 mg daily compared with placebo at 24 and 48 weeks after start of treatment relative to baseline. For definition of ‘retention’ and ‘maintenance’ see text.
The impact of the currently available hormonal therapies on sexual interest and function varies, and therefore may be an important consideration when selecting therapy for specific patients with advanced disease. Loss of libido and sexual function are frequently experienced after medical or surgical castration. The data presented in this review confirm the widespread perception that monotherapy with a non-steroidal antiandrogen offers benefits over castration with respect to preservation of libido and sexual potency. Overall, approximately two-thirds of patients with locally advanced or metastatic prostate cancer retain sexual interest following treatment with non-steroidal
antiandrogens, a significantly greater proportion than after medical or surgical castration.
Figure 5 Sexual function as assessed by the GRISS (Golombok-Rust Inventory of Sexual Satisfaction).
Sexual function is also maintained to a greater extent than following castration. These findings are consistent for the two most studied non-steroidal antiandrogens (bicalutamide and flutamide), in groups at different disease stages (locally advanced and metastatic), in both open and blinded studies, and using different methods of evaluation. Bicalutamide 150 mg/day has been the most extensively investigated non-steroidal antiandrogen, and findings on sexual interest are consistent in patients with both locally advanced and metastatic prostate cancer.
In a placebo-controlled study in early, nonmetastatic prostate cancer, a slightly smaller fraction of patients treated with bicalutamide 150-mg monotherapy maintain, or retain some, sexual activity and function after 48 months when compared with placebo. However, the difference is small. Longer followup seems warranted. Bicalutamide appears to be a useful treatment option for men with advanced prostate cancer who wish to remain sexually active.